HOW TO BUILD A SUSTAINABLE FRANCHISE
As a consequence, we have seen that many pharmaceutical companies radically shift their R&D activities – a decision that will determine the future success of a company to a high extend. Defining a therapeutic area strategy, therefore, has to follow a systematic funnel identifying attractive segments from a company’s perspective. A multi-stage process is necessary to deduce the new TA strategy.
stradoo will ensure such approach being successful when leveraging all available sources of knowledge within a company and making best use of additional data and external expert support.
We have seen that many pharmaceutical companies radically question and shift their R&D activities. Companies do not only withdraw from traditional research areas, they even break with their roots and explore new, promising frontiers of research.
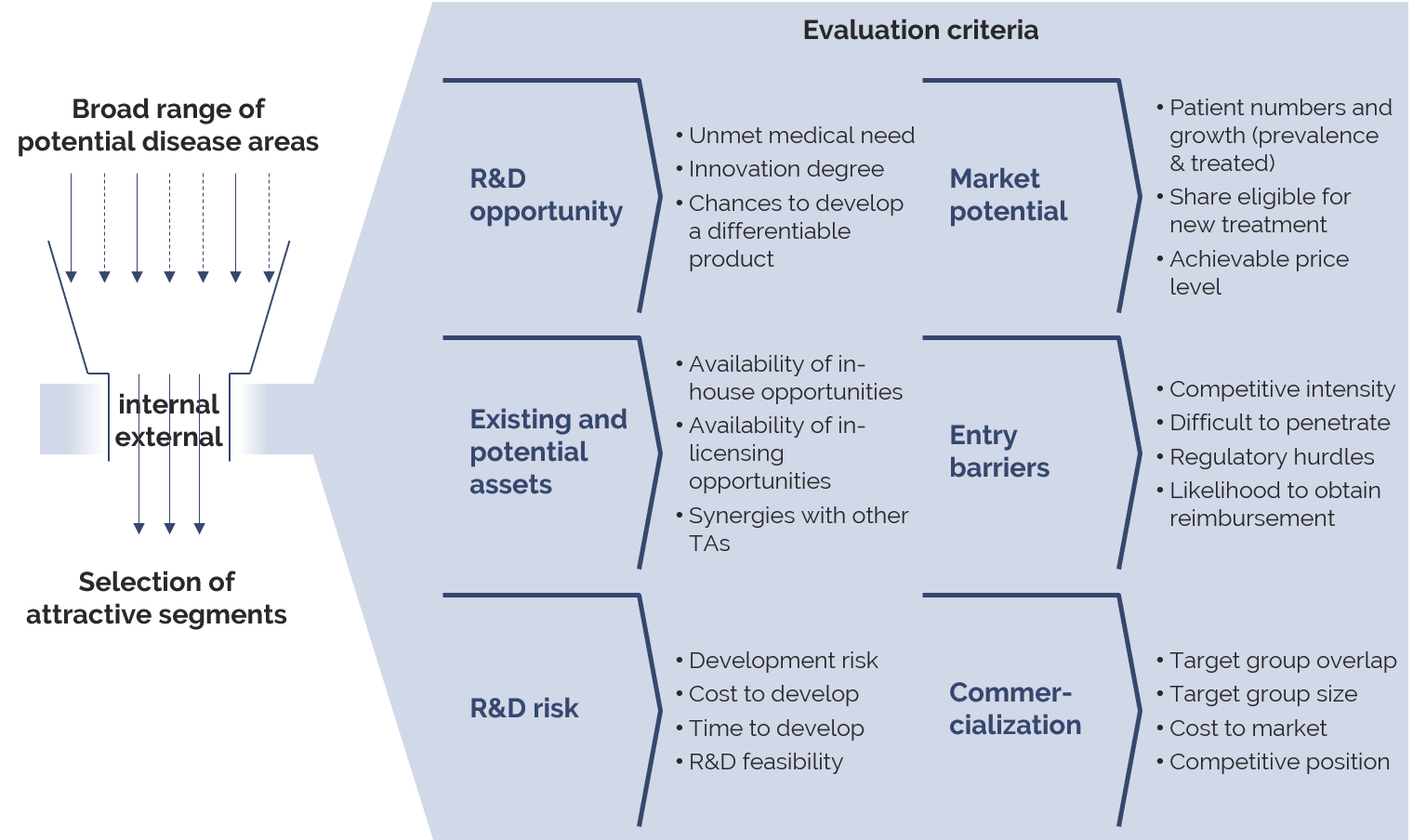
stradoo believes that defining a TA strategy today has to follow a systematic funnel to identify attractive segments from a company’s perspective. As illustrated by the figure below, a multi-stage process is necessary to de- duce the new TA strategy of a pharmaceutical company. Targets with high potential are identified by systematic segmentation, a detailed analysis of the “unmet medical needs” and the market environment.
Segmentation
As a first step, all potentially interesting therapeutic segments have to be selected and brought into a systematic segmentation. An in-depth understanding of the existing indications, clinical endpoints and notably of the “unmet medical need” within the therapeutic area needs to be developed.
Medical and market analyses
As a consequence of setting the R&D focus of a company on certain diseases or indications, all R&D resources of the company are aligned to the chosen focus. Before such a decision can be made – a decision that affects the work of hundreds of employees over many, many years – the scientific and economic potential of an indication has to be analyzed. As soon as the scope of segments is defined within the context of the segmentation process, a detailed medical and market analysis has to be elaborated.
On top of the purely scientific analysis of the potential, companies have to question at a very early stage, what a potential commercialization would look like. What target group should be addressed? What internal structures are needed for a successful commercialization? What regional differences have to be considered? What does the future price-level of the targeted therapy look like?
Attractiveness evaluation
stradoo´s attractiveness evaluation consists of a comparative analysis, visualized in attractiveness matrices. Segments and targets are thereby evaluated with regard to their attractiveness along several criteria.
Such criteria include the unmet medical needs, the number of available targets within an indication and the R&D risk, but also commercial aspects such as number of treated patients, achievable price level, market access hurdles and market commercialization requirements. Combining the different attractiveness criteria will identify and visualize the segments on which to base the corporate strategy.
Strategic options and option quantification
Finally, strategic options have to be developed that quantify the future of the company financially. This means, comprehensive strategic options are built, based on the identified targets, and quantified in form of business cases. The business cases need to describe in detail what in-licensing cost and roy- alties have to be considered, how high the preclinical and clinical R&D investments will be, what finan- cial resources will be required for market commercialization, what co-marketing or co-promotion agreements will be made and how these will affect the cost structure of the strategic option and last but not least, how the therapy price will develop over the next decades, following market entry.
In addition to the strategic options, scenarios that depict non-controllable, external influences have to be developed. These scenarios serve as a foundation for future decisions. They allow for the simulation of the statistical risk (success rates in dependence of clinical phases and milestones achieved), as well as the competitors’ behavior, market developments and price variability.
It is evident that the definition of “Therapeutic Area strategy” involves juggling quite a few unknown variables. As unpredictable as pre-clinical and clinical development is, it is highly important, that all variables and framework conditions applied in the development of a business case should be described in detail.
stradoo will guide your organization through those steps provide insights in science and tools to execute.



